The Global Fight Against Malaria
SANGER INSTITUTE CASE STUDIES

Malaria is a significant global healthcare challenge
Despite much progress that has been made to battle the disease, it remains a significant global healthcare challenge. By the 1970's, through public health measures such as the use of insecticides and anti-malarial drugs, the disease was virtually eliminated from the more affluent regions of the globe, but sadly it remains endemic in most of the tropical and subtropical regions of the world today, disproportionately affecting poor and vulnerable populations. It is found in more than 100 countries mainly in tropical regions of the world, including: large areas of Africa and Asia, Central and South America, Haiti and the Dominican Republic, parts of the Middle East and some Pacific islands. In regions where transmission remains high, it is often the leading cause of illness and death.
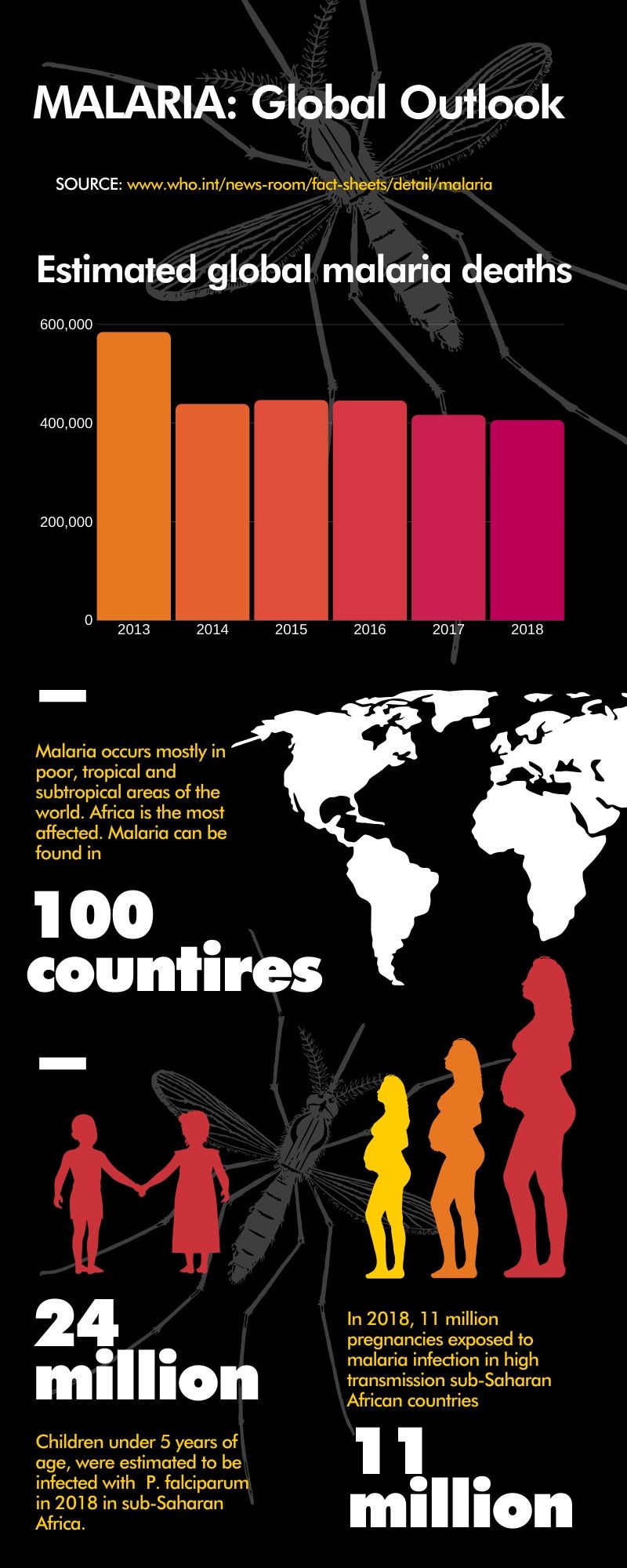
Figures for the infographic taken from: www.who.int
Figures for the infographic taken from: www.who.int
The Challenge
Malaria is very effective at evading treatment
Why is it difficult to treat malaria?
Malaria has a complicated life cycle and often stays undetected for most of it, divides rapidly before causing any symptoms, and spreads through its host, reproducing very quickly. This creates many opportunities for the disease-causing parasite to develop resistance. Any genetic mutation that allows a mosquito or parasite to survive insecticide or drug treatment, respectively, passes onto the next generation, over and over, until most of the surviving population also becomes resistant.
The disease life cycle
Malaria is caused by parasites that live, mate and sexually reproduce inside female mosquitos. These mosquitoes can lay over 500 eggs before dying, and in their short life-cycle of just 8-10 days, the mosquitos infect humans by biting them and transmitting the parasites. Once inside the bloodstream, the parasites travel to the human liver where they multiply relatively slowly without causing any symptoms. When they eventually leave the liver to infect red blood cells, they divide much more rapidly by bursting out of red blood cells, infecting more blood cells by doing so. Left untreated, the number of parasites eventually numbers in billions or trillions and infected individuals finally show symptoms such as a high temperature, muscle pains, diarrhoea, vomiting, headaches and feeling shivery.
We need better vaccines, drugs and disease surveillance
To boost current efforts to battle malaria, we need to develop new tools such as vaccines that prevent transmission, new drugs that can target the disease in the liver (before it destroys blood cells) and investment in systems that make existing tools more effective and economical.
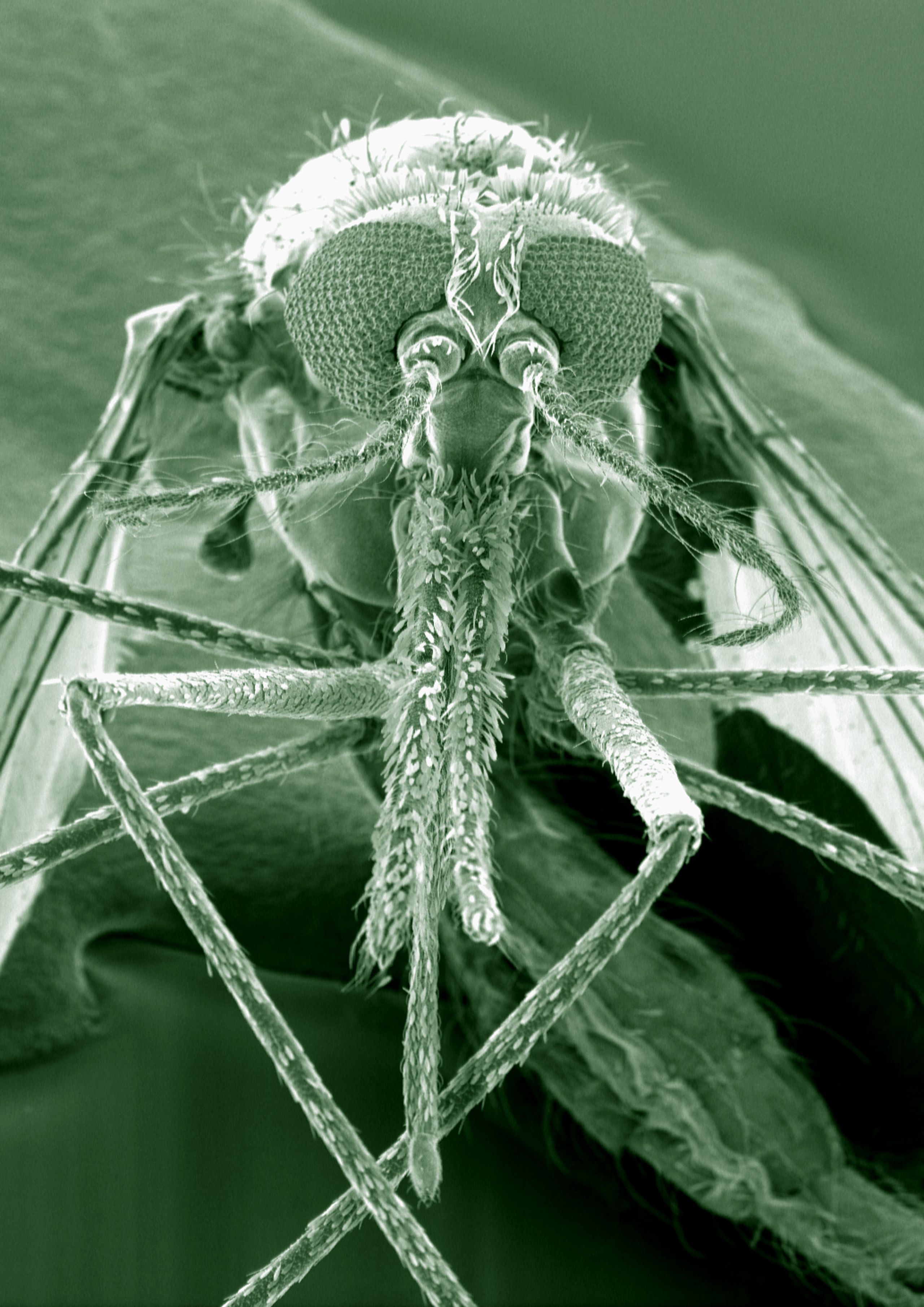
This (SEM) shows the Anopheles gambiae mosquito. The species is thought to be the principal vector of malaria in Africa. Kevin Mackenzie, University of Aberdeen. Attribution 4.0 International (CC BY 4.0)
This (SEM) shows the Anopheles gambiae mosquito. The species is thought to be the principal vector of malaria in Africa. Kevin Mackenzie, University of Aberdeen. Attribution 4.0 International (CC BY 4.0)
Three Sanger projects to help fight malaria
1. New targets for drugs | 2. A cost effective vaccine | 3. Better disease surveillance

Sanger's extraordinary science
The Sanger Institute uses genome sequences to advance understanding of the biology of humans and pathogens in order to improve human health. The Institute has the largest concentration of genomics experts in the world, with 25 years of experience, working across five distinct areas of scientific focus. Our researchers work towards the application of Sanger's science in a number of areas such as drug discovery, vaccine discovery, epidemiological surveillance, diagnostics and precision medicine. The ethos at Sanger demands that infectious diseases, such as malaria, currently afflicting the developing world receive equal attention and resource to the diseases of affluence such as cancer and type 2 diabetes.
Applying science through translation
Discoveries made in the lab, usually require further development before they will attract enough funding for improvement and deployment at the scale needed to affect meaningful societal impact. Technology transfer activities at Sanger are sophisticated, well-funded and thriving; offering support, facilities, access to funding, partnership opportunities and an incubator style space for early stage spin-outs or startups. Between 2012 and 2017, the Sanger Institute's Technology Transfer Team backed three ambitious projects tackling areas key to eliminating malaria.

CASE STUDY #1
Finding new markers to help make better medicines
Targeting parasites in the liver stage

The search for new targets for anti-malarial drugs
Because the malaria parasites divide quite slowly whilst still in the liver, that is the stage at which they have less opportunity to develop a resistance to any potential drugs. Finding a way to kill them before they leave the liver would prevent infected individuals from falling ill. However, developing anti-malarial drugs is still difficult due to the lack of model systems to genetically identify drug targets for the liver stage of the parasite.
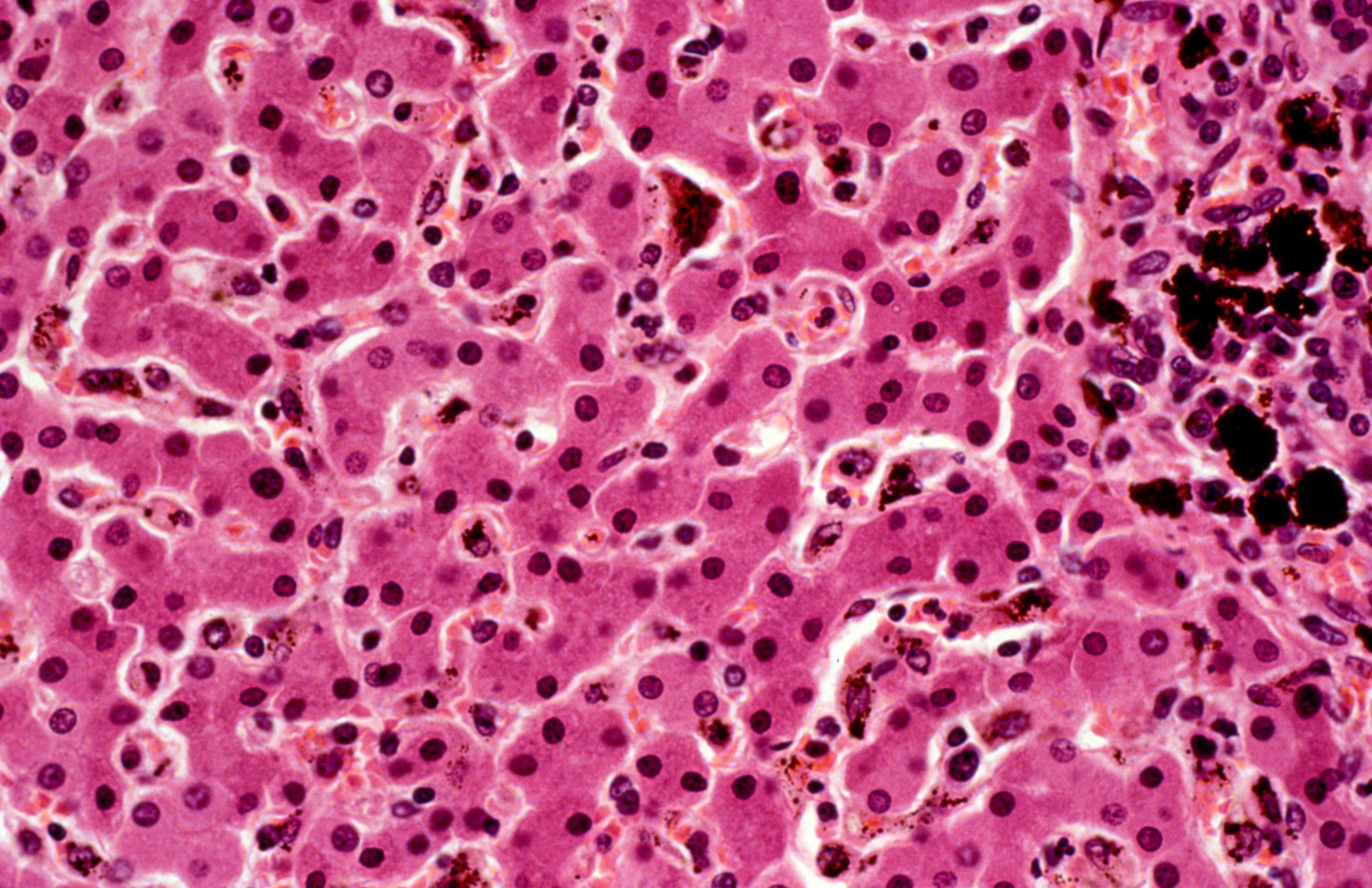
This high power microscopic view of a section of liver from an autopsy of a child with falciparum malaria shows haemozoin deposition. H&E stain. Credit: Plasmodium falciparum malaria: liver. Wellcome Collection. CC0 1.0 Universal
This high power microscopic view of a section of liver from an autopsy of a child with falciparum malaria shows haemozoin deposition. H&E stain. Credit: Plasmodium falciparum malaria: liver. Wellcome Collection. CC0 1.0 Universal
Oliver Billker and his team at Sanger have developed a technique to help genetically identify the targets of drugs on parasites in the blood. This work (published in 2015) provided valuable insights into other compounds that might work against the parasites, other areas of vulnerability, and potential mechanisms of resistance.

Pictured: The Billker Research Group. The Group is now based at The Laboratory for Molecular Infection Medicine Sweden (MIMS) within the Nordic EMBL Partnership for Molecular Medicine (http://www.mims.umu.se/groups/o-billker.html).
Pictured: The Billker Research Group. The Group is now based at The Laboratory for Molecular Infection Medicine Sweden (MIMS) within the Nordic EMBL Partnership for Molecular Medicine (http://www.mims.umu.se/groups/o-billker.html).
Funding the research to help secure industry collaboration
Realising the value of the application of this research in the discovery of new targets for drugs that could be used on parasites in the liver stage, the Sanger Institute's Technology Transfer Team helped to support the work of the Bilker team through funding for almost a whole year which enabled the scientists to continue their work, and led to a further grant from the Open Lab Foundation. The team is currently using the combined resource and expertise of its new partners to find potential new drugs against malaria.
CASE STUDY #2
Developing a cost-effective vaccine
To lower infection rates and help prevent transmission
A new vaccine target
In spite of many efforts, researchers have struggled to find a good target for vaccination against the family of parasites that causes malaria. These genetically diverse parasites often change shape every time they enter a new stage of their life cycle. However, in 2011, Gavin Wright’s team at the Sanger Institute identified a protein known as pfRH5 which the parasites absolutely require for the invasion of red blood cells. Unlike most other plasmodium proteins involved in infecting blood cells, pfRH5 looked the same across all strains known to cause malaria, making it an excellent vaccine target.

Pictured: The Wright Group, Wellcome Sanger Institute
Pictured: The Wright Group, Wellcome Sanger Institute
Lowering vaccine development costs
On the back of these results, the Sanger Institute's Technology Transfer Office helped to support a project focusing on purifying a small, soluble portion of the protein that could still effectively elicit an immune response. In theory, vaccine manufacturers could then produce a fragment of the protein more cheaply than the entire protein, which is key for tackling a disease that primarily affects the poorer countries.
Unlocking funding to help the technology reach the clinic
Given this rare opportunity to progress a technology into the clinic, for so widespread a disease and based on other encouraging data to support further work towards producing biochemically active RH5 in a cost-effective way, the Sanger Institute's Technology Transfer Office supported this initiative by helping the Wright team to secure funding. While the results from this project did not produce a protein with the desired activity, current clinical trials that were developed using the full-length pfRH5 protein have shown promising results thus far.
“There is a great need for an effective malaria vaccine, and the RH5 complex is the most important link between parasite and host that we yet know of. Our study shows us how this works, and reveals other essential malarial proteins to target. As RH5 is only exposed from the parasite briefly, a combination vaccine based on P113, RH5 and other proteins in the complex could be more effective than RH5 alone.”

CASE STUDY #3
Creating better surveillance systems
To track genetic changes in the disease-resistant parasites and mosquitos
New computational tools to track and understand disease evolution
In 2008 following a study conducted in Cambodia, Dominic Kwiatkowski's Group and collaborators discovered that the most lethal of known malaria parasites, P. falciparum, had started responding much more slowly to front-line combination treatment DHA-PPQ (dihydroartemisinin and piperaquine). We now know that until 2013, resistance had spread under the radar and continued use of DHA-PPQ likely hastened this process. Evolutionary change and adaptation allows malaria to develop resistance to current treatments. Understanding how this occurs, and quickly, will help us find ways to manage the disease more effectively.

Pictured: The Kwiatkowski Group, Wellcome Sanger Institute
Pictured: The Kwiatkowski Group, Wellcome Sanger Institute
Alongside research dedicated to targeting vulnerabilities in the malaria infection cycle, Dominic Kwiatkowski’s group is also developing cutting edge genetic and computational tools to track evolutionary changes in mosquitos and the malaria parasites.
“There is now an urgent need to provide national malaria control programmes with the tools for active genomic surveillance that will help to detect new emergences of resistance as soon as they arise and thereby reduce the risk of a major global outbreak.”
Translating research into a funded stand-alone project
To help the Kwiatkowski group demonstrate the levels of reliability, accuracy, sensitivity, speed, cost-effectiveness and scalability required for their platform and methodology to be adopted as a public health surveillance tool, they needed financial sponsors. The Sanger Technology Transfer Team helped the researchers to secure financial backing to continue their work.
The project was completed successfully, and becoming known as the SpotMalaria Project, it now operates in several global malaria hotspots using technology to monitor drug resistance.

Wellcome Sanger Institute sequencing labs, Wellcome Genome Campus
Wellcome Sanger Institute sequencing labs, Wellcome Genome Campus

Blood spot test for malaria in Cambodia
Blood spot test for malaria in Cambodia
Partner with Sanger
translation@sanger.ac.uk
| +44(0)1223 834244

More about Sanger Innovations
Find out more about our other translation success stories by clicking on the images below.



